- Nickel Dmg Complex Structure Crossword
- Dimethylglyoxime Nickel (ii) Complex Structure
- Nickel Dmg Complex Structure Definition
- Nickel Dmg Complex Structure List
- Nickel Dmg Complex Structure Chart
- Nickel-dimethylglyoxime Complex Structure
Properties: Scarlet-red, cryst powder. Sublimes at 250°. Insol in water, acetic acid, ammonia. Sol in dil mineral acids and appreciably sol in abs alcohol. DmgH 2 is used in the analysis of palladium or nickel. Its coordination complexes are of theoretical interest as models for enzymes and as catalysts. Many related ligands can be prepared from other diketones, e.g. Dimethylglyoxime can be prepared from butanone first by reaction with ethyl nitrite to give biacetyl monoxime. Metal ions in solution are surrounded by an array of ions or molecules called ligandsto form coordination complexes. Those of main group metal ions such as Na(I), Hg(II) or Al(III) (and Cu(I)), which have no lone-pair electrons, have the high symmetry shapes that minimise the ligand-ligand repulsions and at the same time, in the covalent picture, the repulsions between the bonding pairs of electrons.
| Dimethylglyoxime | |
|---|---|
2,3-Butanedione Dioxime | |
Dimethylglyoxime, Diacetyl dioxime, Chugaev's Reagent 2,3-diisonitrosobutane | |
| Identifiers | |
| CAS number | 95-45-4 |
| ChemSpider | 10606175 |
| UNII | 2971MFT1KY |
| RTECS number | EK2975000 |
| Jmol-3D images | Image 1 |
| |
| |
| Properties | |
| Molecular formula | C4H8N2O2 |
| Molar mass | 116.12 g mol−1 |
| Appearance | White/Off White Powder |
| Density | 1.37 g/cm3 |
| Melting point | 240-241 °C, 513-514 K, 464-466 °F |
| Boiling point | decomp. |
| Solubility in water | low |
| Structure | |
| Dipole moment | 0 |
| Hazards | |
| MSDS | External MSDS |
| R-phrases | R20/22 |
| S-phrases | R22, R36/37 |
| Main hazards | Toxic, Skin/Eye Irritant |
| NFPA 704 | 0 0 |
| Related compounds | |
| Related compounds | Hydroxylamine salicylaldoxime |
| (verify) (what is: /?) Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
| Infobox references | |
Dimethylglyoxime is a chemical compound described by the formula CH3C(NOH)C(NOH)CH3. This colourless solid is the dioxime derivative of the diketone diacetyl (also known as 2,3-butanedione). DmgH2 is used in the analysis of palladium or nickel. Its coordination complexes are of theoretical interest as models for enzymes and as catalysts. Many related ligands can be prepared from other diketones, e.g. benzil.
Preparation
Dimethylglyoxime can be prepared from butanone first by reaction with ethyl nitrite followed b hydroxylamine monosulfonate:[1]
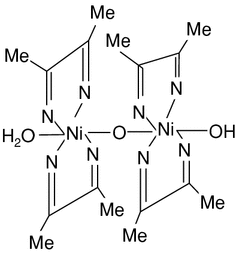
Ni(dmgH)2
Structure of Ni(dmgH)2
Nickel Dmg Complex Structure Crossword
A sample of Ni(dmgH)2
Dimethylglyoxime Nickel (ii) Complex Structure
Dimethylglyoxime is used as a chelating agent in the gravimetric analysis of nickel. The use of DMG as a reagent to detect nickel was discovered by L. A. Chugaev in 1905.[2] For qualitative analysis, dmgH2 is often used as a solution in ethanol. It is the conjugate base, not dmgH2 itself, that forms the complexes. Furthermore, a pair of dmgH- ligands are joined through hydrogen bonds to give a macrocyclic ligand. The most famous complex is the bright red Ni(dmgH)2, formed by treatment of Ni(II) sources with dmgH2. This planar complex is very poorly soluble and so precipitates from solution. This method is used for the gravimetric determination of nickel, e.g. in ores.
Cobaloximes
The nitrogen atoms in dmgH2 and its complexes are sp2 hybridized.[3] Because of the planarity of the resulting ligand, the macrocycle [dmgH]22- resembles some biologically important macrocyclic ligands, as found for example in vitamin B12 and myoglobin. A well known family of model complexes, the cobaloximes, have the formula CoR(dmgH)2L, where R is an alkyl group and L is typically pyridine. In such complexes, L and R occupy “axial” positions on the cobalt, perpendicular to the plane of the (dmgH)2.
References
- ^Semon, W. L.; Damerell, V. R. (1943), 'Dimethylglyoxime', Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv2p0204; Coll. Vol.2: 204
- ^Lev Tschugaeff (1905). 'Über ein neues, empfindliches Reagens auf Nickel'. Berichte der deutschen chemischen Gesellschaft38 (3): 2520–2522. doi:10.1002/cber.19050380317.
- ^Girolami, G.. S.; Rauchfuss, T.B.; Angelici, R. J. (1999). Synthesis and Technique in Inorganic Chemistry: A Laboratory Manual (3rd ed.). pp. 213–215.
Nickel Dmg Complex Structure Definition
Nickel Dmg Complex Structure List
Wikimedia Foundation. 2010.
Nickel Dmg Complex Structure Chart
Look at other dictionaries:
Nickel-dimethylglyoxime Complex Structure
- diméthylglyoxime — ● diméthylglyoxime nom féminin Dénomination courante de la dioxime du diacétyle. (C est un réactif analytique, employé pour le dosage du nickel, du palladium et du bismuth.) … Encyclopédie Universelle
- dimethylglyoxime — di·meth·yl·gly·ox·ime (di meth″il gli okґsēm) a reagent for nickel and similar metals; see dimethylglyoxime test, under test … Medical dictionary
- dimethylglyoxime — dimetilglioksimas statusas T sritis chemija formulė [CH₃(HON=)C]₂ atitikmenys: angl. dimethylglyoxime rus. диметилглиоксим ryšiai: sinonimas – 2,3 butandiono dioksimas … Chemijos terminų aiškinamasis žodynas
- dimethylglyoxime — noun The oxime 2,3 butanedione dioxime that is used as a reagent in the analysis of nickel and palladium … Wiktionary
- dimethylglyoxime — di·meth·yl·glyoxime … English syllables
- dimethylglyoxime — noun a compound used in analysis as a precipitant for palladium or nickel (Freq. 2) • Hypernyms: ↑precipitant * * * “+ noun Etymology: International Scientific Vocabulary dimethyl + glyoxime : a crystalline compound CH3C(NOH)C( … Useful english dictionary
- dimethylglyoxime test — (for nickel) the object or substance being tested is placed in a solution of dimethylglyoxime; if nickel is present, the solution will turn brown … Medical dictionary
- Synthese de complexes du nickel — Synthèse de complexes du nickel Nous désirons synthétiser 3 différents complexes du nickel dont le nombre d’atomes coordonnés varient de 4 à 6 ainsi que leurs propriétés optiques et magnétiques. Sommaire 1 Principe 2 Schéma de la synthèse 3 Mode… … Wikipédia en Français
- Synthèse de complexes du nickel — Nous désirons synthétiser 3 différents complexes du nickel dont le nombre d’atomes coordonnés varient de 4 à 6 ainsi que leurs propriétés optiques et magnétiques. Sommaire 1 Principe 2 Schéma de la synthèse 3 Mode opératoire … Wikipédia en Français
- Диметилглиоксим — Общие Химическая ф … Википедия
